Sickle cell disease is a devastating chronic genetic disorder caused by inheritance of sickle heamoglobin gene which is reported to results to various clinical manifestations such as vaso-occlusive crisis, heamolysis, anaemia, risk of infection and other complications [1]. This genetic defect is said to be due to a substitution of Glutamic acid, a hydrophilic amino acid with valine, which is hydrophobic, at the sixth position of the beta-globin chain [2]. Heamoglobin S (HbS), the heamoglobin that is produced owing to this abnormality, is known to be poorly soluble and polymerizes when deoxygenated [3]. On the other hand Oxidative stress is known to plays a very critical role in the pathophysiology of SCD and its complications [4-5]. Generally, Oxidative stress is defined as an imbalance between oxidants/free radical and antioxidants [6]. Several molecular mechanisms have been proposed to contribute towards an increased oxidative burden in sickle cell patients. However for this research, the excessive level of Fe2+ radicals due to free heamoglobin and the impaired bioavailability of endogenous nitric oxide in SCD were assessed.
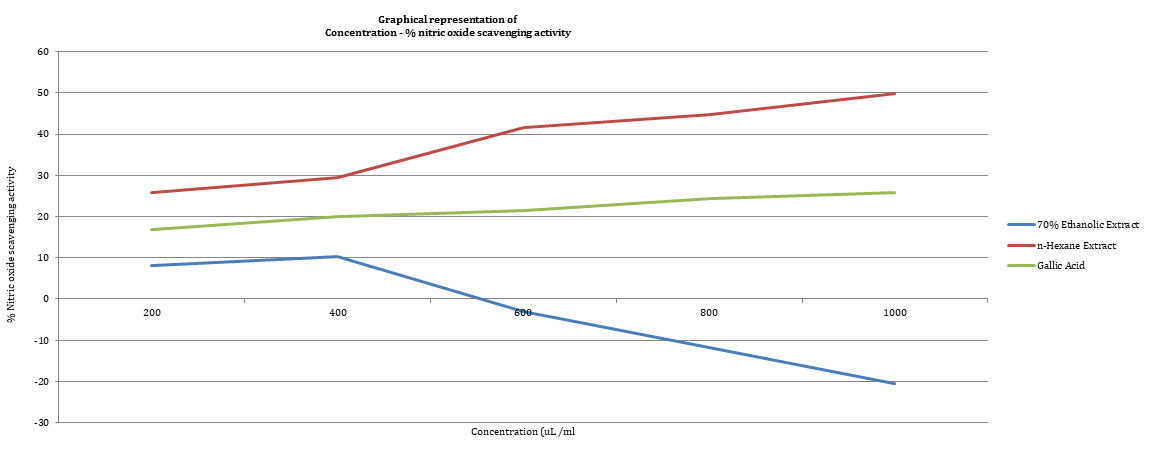
Nitric Oxide (NO) imbalance in SCD is known to be responsible for the pain crisis in a sickle cell carrier. NO released from the endothelium is a potent endogenous vasodilator that acts by reducing intracellular calcium concentration in smooth muscle through a cascade of events therefore producing relaxation and dilation of the vascular smooth muscles and increases blood flow through the blood vessels [7-9]. In addition, nitric oxide also suppresses platelet aggregation and promotes expression of cell adhesion molecules on endothelial cells [10-13] .
Recent studies have demonstrated that patients with sickle cell disease suffer from impaired bioavailability of the endogenous vaso-dilator, nitric oxide and this could be due to reduced plasma L-arginine level, scavenging of nitric oxide by cell-free plasma heamoglobin and by reactive oxygen species [14-18].
There are several methods which are being used widely in biological system for assay of NO; however, the Griess reaction assay as demonstrated by Johann Peter Griess in 1879, was used for this research work due to its simplicity and low cost.
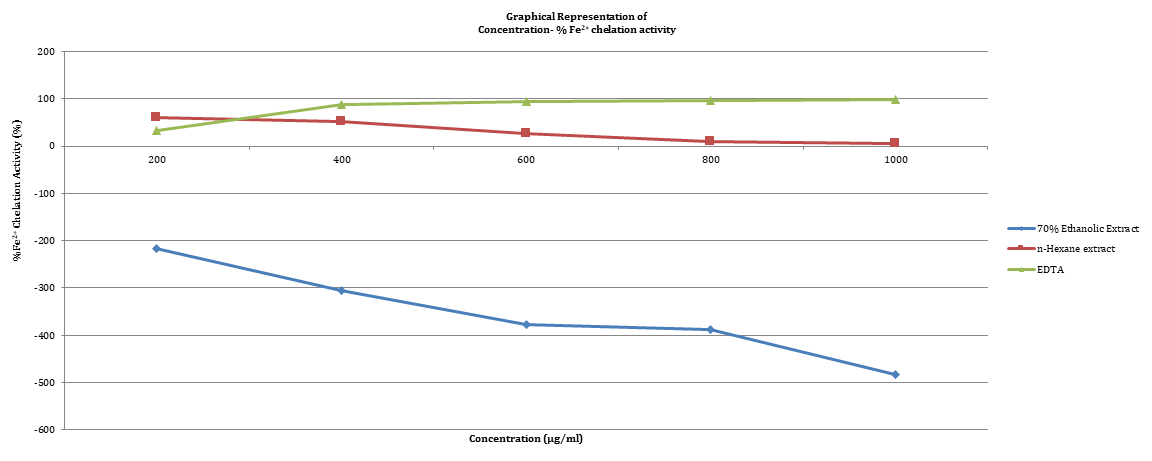
A typical human red blood cell is known to contain about 270 million heamoglobin with each heamoglobin having four heme biomoiety. Bridges, 2021 reported that red blood cells of an average adult male collectively store about 65 % of the total iron contained in the body. Old and structurally defective erythrocytes are destroyed by the phagocytic cells of the reticuloendothelial system in the spleen, liver, bone marrow and lymph node, a process known as extravascular heamolysis [19], therefore, only little heamoglobin escapes into the plasma. In contrast to degradation of normal erythrocytes, sickle red blood cell is known to survive for just 10 – 20 days as observed by Sebastiani et al, 2007 and is majorly destroyed intravascularly, a phenomenon known as “intravascular heamolysis”[11]. Intravascular heamolysis results to the availability of cell-free plasma heamoglobin, which mop up nitric oxide about 1000-fold more rapidly, consequently reducing the bioavailability of nitric oxide significantly in patients with sickle cell disease [11].
Furthermore, the issue of Iron overload is a clinically significant and inevitable complication in transfusion, especially in patients with thalassemia [20-21]. The excess iron is stored in the heart, liver and pancreas and can lead to poisoning of these organs, consequently resulting in conditions such as cancer, liver cirrhosis and irregular rhythm of the heart [20]. Therefore, the use of an iron chelators therapy is of great importance in the treatment of transfusion- dependent iron overload and in maintenance of the normal body iron levels in tissues [22].
The ethnomedicinal use of natural products in bid to manage or treat SCD could be as old as when the disease was discovered, though only relatively few have been validated scientifically. Among the scientifically validated herbal remedies used ethnomedicinally in management of SCD, the reddish brown freshly fallen leaves of Terminalia catappa was further analysed in this research.
Terminalia catappa is a large tropical tree that belongs to the Combretaceae family. Its common names include country almond, Indian almond, Malabar almond, sea almond, tropical almond, beach almond and false kamani [23]. In 2014, Praveena reported that country almond contains various phytochemicals such as steroids, carbohydrates, alkaloids, triterpenes, saponins, polyphenols, flavonoids, glycosides and tannins.
Aside its ethnomedicinal use in management of SCD, the leaves of Tropical almond have also been demonstrated to have significant antibacterial activity [24], anti-ulcer properties [25], aphrodiastic properties, antifungal properties [26], anti-oxidative properties [27], antinociceptive properties [28], antimicrobial activity [29], anti- aging properties [30], antidiabetic properties [31] and anti-inflammatory properties [32]. It has been shown to have therapeutic effects on hepatitis [32] and has been used ethno-medicinally in treatment of skin diseases such as dermatitis.
In 2003, Moody et al demonstrated that the freshly fallen reddish brown leaves of Terminalia catappa, L. Family Combretaceae, exhibit antisickling effect; Mgbemene and Ohiri [33] had also demonstrated the ability of the ethanolic extract of the leaves of T. catappa to inhibit osmotically-induced hemolysis of human RBCs in a dose-dependent manner. They also illustrated the ability of T. catappa leaves to prolong the clotting time of uncoagulated blood and the effectiveness of a 1.0 mg/ml solution of the extract in preventing and reversing the sickling of sodium meta-bisulphite induced sickled RBCs. Furthermore, Samuel et al. [34] validated the scientific basis for the application of the methanolic extract of T. catappa leaves in the management of sickle cell anaemia in traditional medicine.