The method used for sampling and laboratory analysis are according to the America Standard Testing Material (ASTM), American Petroleum Institute (API) and Petroleum Institute of India (PII). The parameters monitored for toxicity levels of refinery effluents are; pH, Temperature, Electrical conductivity, Oil and grease, Total dissolved solids, Total suspended solids, Biochemical oxygen demand, Chemical oxygen demand, Cyanides, Salinity, Metals: Nickel, Lead, Manganese, Cadmium and Chromium
2.6 Parameters of Pollution Monitored in Refinery Effluent
Pollution parameters are generally used for characterizing the various forms of pollutants. The parameters are largely dependent on the sources of effluents whereas domestic sewage contains impurities. These parameters of pollution in refinery effluents can be classified under physical, chemical and microbial qualities.
2.6.1 Physical Parameters
This are generally reported in terms of temperature, colour, odour, turbidity and pH
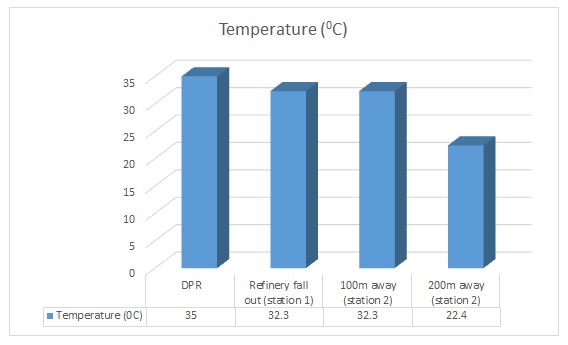
Temperature: The temperature of the water samples monitored is in degree centigrade. The temperature of the refinery wastewater in warm climate is slightly lower than air temperature during most of the year and it has effect on microbial activities.
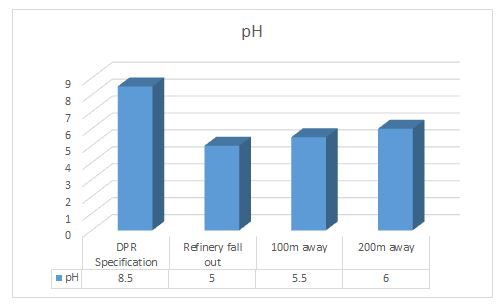
pH: This is the negative logarithms base 10 of hydrogen ion concentration(pH=-log10[H+]). The pH of refinery wastewater was monitored with a pH meter and made sure it was accordance with the international standard before being discharged.
Turbidity: This is caused by a variety of suspended and colloid solids. The turbidity is expressed in the form of total dissolve solids and conductivity.
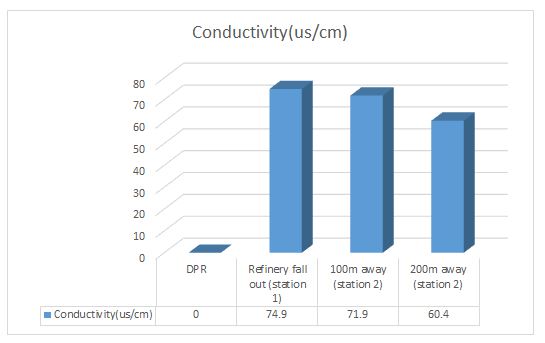
Electrical conductivity and resistivity of water: The unit electrical conductivity is Siemens per centimeter. The actual resistance of the cell Rx is measured in Ohms. The conductance 1/Rx is directly proportional to the length of the path L (cm). 1/Rx=K*A/L where K is the conductivity and expressed in millisiemens/centimeter at a specified temperature normally at 25oC.
Electrical Resistivity: Resistance in Ohms measured between opposite faces of centimeter cube of an aqueous solution at specified temperature[Rx=R*L/A]. The values are usually expressed in Ohm centimeter at a specified temperature of 25oC.
2.6.2 Chemical Pollution Parameters
In refinery waste, there are mainly organic and inorganic constituents including fat and grease. The constituents are stable and decomposed slowly by microorganisms. Organic constituents are monitored by Biochemical oxygen demand(BOD) and Chemical oxygen demand(COD), phenolic compounds and cyanides. Inorganic parameters are acidity and alkalinity, chloride, phosphate, ammonia and heavy metals. All these are monitored to make sure that they are within FEPA specifications before they are discharged into sea.
2.7 Laboratory Analysis
2.7.1 Determination of Total Suspended Solids in Water
Summary of method: The total matter is determined by evaporation of an appropriate aliquot or the particulate are separated by filtration and dried and weighed.
Method: 100mL of water samples was measured and filtered through 0.45µm filter membrane. Before filtration, the filter paper was dried in oven at 105oC and cooled in the desiccator and weighed. The particulate matter was dried and weighed.
Calculation:
Total suspended solids= 
ie
Result expressed in ppm or mg/L
Significance: It is important for biologically treated effluents and for many industrial wastes. The suspended matter is largely organic and thus responsible for significant proportion of the oxygen demand. Thus if discharged to steam it would consume undesirable amount of dissolved oxygen.
2.7.2 Determination of Oil and Grease
Scope: This is to determine the oil and grease in water by gravimetric method.
Procedure: 200mL of the wastewater sample was placed in 300mL separating funnel and acidified with 5mL of conc. HCl. 40mL of CCl4 was added and shaken vigorously for extraction. The pressure was always released at intervals; the 100mL beaker was dried and weighed. The CCl4 layer was filtered though a filter paper containing anhydrous sodium sulphate into a known weight 100mL beaker. The anhydrous sulphate is to remove the traces of water from oil. The beaker and the contents were placed on a hot plate in a fume cupboard evaporated to dryness. The beaker was removed and cooled in a dessicator for 30mins before reweighing.
Calculation:
Oil/grease content=
ie
Significance: The presence of oil and grease in domestic and industrial wastewater is of concern to the public because of its deleterious aesthetic effects and its impacts on aquatic life. Regulation and standards have been established that requires monitoring of oil and grease in wastewater. Oil and grease standard in water is 0.5ppm.
2.7.3 Determination of Phenolic Compounds by Direct Colorimetric Method
100mL of the distillate was transferred to 100mL volumetric flask or suitable aliquot diluted to 100mL. Blank was prepared with 100mL of distilled water measured into 100mL volumetric flask. 5mL of NH4OH solution was added to the 100mL distillate and adjusted the ph 9.8-10.2 with NH4OH. 2mL 0f 4-amino antipyrine and 2mL of Potassium ferric cyanide was added and mixed vigorously. The mixture was left for 15mins for colour development. The mixture was later scan at 510nm with10mm absorption cell. The colour intensity is proportional to the amount of the phenolic compounds present in the sample.
Calculation: Conc. from UV dillutor factor
dillutor factor
=Phenol mg/L or ppm
2.7.4 Determination of Cyanide
Scope: The method covers the determination of total cyanide in water and saline water
Significance: The presence of cyanide in industrial, domestic and surface waters is of concern because of its toxicity.
Summary of methods: This is based on the decomposition of nearly all of the metalo-cyanide complexes and simple cyanides from a strongly acidified sample during a one hour reflux distillation procedure. To ensure the breakdown of the very tightly complexed iron cyanide compounds; a decomposition catalyst magnesium chloride is added to the sample before distillation. The cyanide content of the absorption solution is determined colourimetrically.
Procedure: 250mL of water sample were collected in 500mL beaker and 20mL of Zinc acetate into 500mL distillation flask and refluxed for 1hr. The distillation was collected in a 100mL cylinder containing 20mL of sodium hydroxide(0.04N)
The sodium hydroxide absorbed the cyanide and converted to sodium cyanide 10mL 0f the aliquot was collected in 50mL volumetric flask. 4mL of phosphate buffer, 2mL of chloramines T and 5mL of pyridine solution were added into the sample and development and measures with 10mm absorption cell at 578nm.
For blank monitoring, 10mL of 0.04N 9f NaOH was collected in 50mL volumetric flask and other reagents were added and scan at 578nm.
Reagents: Phosphate k buffer, anhydrous sodium, hydrogen phosphate, 3N NaH2PO4.H2O, (138g/L).
Barbatric pyridine preparation: 15g Barbaric into a 250mL volumetric flask and 75mL pyridine and mixed vigorously. 15mL of HCl was added to the mixture and cool to room temperature, then diluted to mark with distilled water and mixed until all the barbituric acid has completely dissolved.
Calculation ppm
µg/50mL=
50/10=Aliquot in 50mL
100mL=Distilled collected
250g-quality of the sample
2.7.5 Determination of Chemical Oxygen Demand using Digital(COD) Meter
Scope: This method describes a procedure for the determination of the total oxidizable material in Refinery effluent and other industrial waste water
Definition: COD is the amount of oxygen consumed under prescribed test condition in the oxidation of organic matter in waste water
Outline: The COD meter is the testing set for COD measurement based on test method of industrial effluent.COD is an index of organic contamination in factory effluent which can ne measured through a simple operation by colometry titration. The PPM is digitally given on the meter.
Principle: A certain quantity of KMnO4(Potassium Permanganate)is added to a water sample and after its acid heating reaction by H2S04 the residual KMnO4 which remains without reacting to the organic matter in the water sample is made to react with Ferrous ions by electro-reduction is detected by an indicator electrode which is directly indicated on the display for COD value. Hence the amount of oxidizable material is directly proportional to KMnO4 consumed.
Reagents: H2SO4/FeNH4(SO4)2: 60g of Ammonium Ferric Sulphate into 400mL of distilled water and 5mL of concentrated sulphuric acid.
N/40 KMnO4 solution: 0.8g of KMnO4 into 1litre of distilled water and store in amber bottle.
Electrode internal solution: Dilute 3.0g KCl into 100mL distilled water.
2.7.6 Determination of Biochemical Oxygen Demand (BOD)
Definition: BOD is the oxygen consumed for biological depredation.
Summary of Method: This is a biological procedure, which attempts to stimulate the natural processes of oxidation of organic matter occurring in a river or stream. The test is carried out by suitably diluting the sample with aerated water and divides the diluted sample between two bottles.
The dissolved oxygen is determined immediately in one bottle and in the second bottle after it has been incubated as a standard temperature of 20oC for 5 days. The period of 5 days is the time at which the rare oxygen consumption would attain a minimum value under natural condition.
Many industrial effluent contain toxic metals phenolic compounds, cyanides which depress the oxidation of organic matter and reducing compound and organic compounds not early amendable to biological oxidation, thus making the BOD test unreliable.
Reagents
Seeding reagents are :
Solution A=FeCl3(0.25g/l)
B=CaCl2(27.5g/1)
C=MgSO4(22.5g/1)
D=K2HPO4(21.75G/1)
KH2PO4(8.5G/1)
Na2HPO4(44.6g/l)
NH4Cl(1.7g/l)
MnSO4(240g/500mL) (solution 1)
KOH 350g, Kl 75g, NaN3 10g/500mL (solution 2)
Na2S2O3.5H2O 6.205g/L
KlO3 3.567g/L
Arrow starch
Procedure: Seeding water was prepared by collecting 5000mL of distilled water and 1% of treated Biodisk water sample added. 5mL of the solution A, 4mL of B, 5mL of C and 5mL of solution D were added to the 5000mL distilled water and aerated for 1hr.
About 3 BOD bottles were used and various dilutions were used for the samples with the seeding water, in each of the bottles containing both samples and seeding water. 1mL of Manganese Sulphate was added to convert the oxygen present at Manganese (iv) oxide and 1ml of (KOH, KI and NaN3) solution was also added to precipitate the sample Manganese hydroxide.
The excess oxygen liberated was then titrated with 0.025N of Na2S2O3 with starch solution as indicator.
Calculation: BOD is calculated as Dissolved Oxygen
Dissolved Oxygen=
BOD= )
)
Where T=Titre value of Na2S2O3
Factor for BOD
20mlLof 1/40N KIO3+5mL of (1:5H2SO4) +Starch solution. Keep in dark for 5mins
Titrate the solution with 1/40N Na2S2O3
N1V1=N2V2
0.025g KIO3×20=0.025g Na2S2O3×V2 Na2S2O3
Factor=20/V2=
For example
To calculate for BOD of an observation pond
Dissolved oxygen=
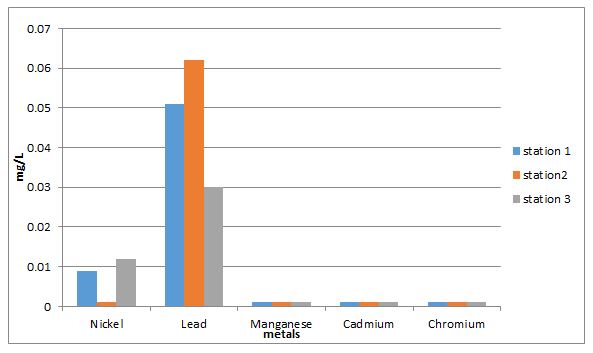
2.7.7 Determination of Heavy Metals
Scope: To determine the toxicity level of heavy metals in the effluent.
Summary of Method: The samples were digested with HCl and into solution with distilled water. The solution was scanned at various wavelengths with Atomic absorption spectrophotometer.
Procedure: The samples was digested with 5mL of concentrated HCl, some water was added and filtered to remove particles. The digested solution was transferred into a 100mL volumetric flask and filled to mark. Different metals where scanned at various wavelengths. The metals observed are: Nickel (Ni) at 248.3nm, Lead (Pb) at 217.0nm, Manganese (Mn) at 279.48nm, Cadmium (Cd) at 228.8nm and Chromium (Cr) at 357.9nm.